
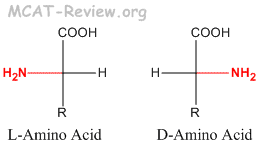
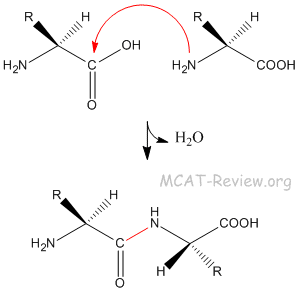
Technically, the main challenges to studying post-translationally modified proteins are the development of specific detection and purification methods. The characterization of PTMs, although challenging, provides invaluable insight into the cellular functions underlying etiological processes. Proteolytic cleavage of peptide bonds is a thermodynamically favorable reaction and therefore permanently removes peptide sequences or regulatory domains.Ĭonsequently, the analysis of proteins and their post-translational modifications is particularly important for the study of heart disease, cancer, neurodegenerative diseases and diabetes. Conversely, phosphatases hydrolyze the phosphate group to remove it from the protein and reverse the biological activity. For example, kinases phosphorylate proteins at specific amino acid side chains, which is a common method of catalytic activation or inactivation. Protein PTMs can also be reversible depending on the nature of the modification. Besides single modifications, proteins are often modified through a combination of post-translational cleavage and the addition of functional groups through a step-wise mechanism of protein maturation or activation. Proteins are also covalently linked to tags that target a protein for degradation. Other modifications occur after folding and localization are completed to activate or inactivate catalytic activity or to otherwise influence the biological activity of the protein. For example, many proteins are modified shortly after translation is completed to mediate proper protein folding or stability or to direct the nascent protein to distinct cellular compartments (e.g., nucleus, membrane). Post-translational modification can occur at any step in the "life cycle" of a protein. Many proteins can also modify themselves using autocatalytic domains, such as autokinase and autoprotolytic domains. These enzymes include kinases, phosphatases, transferases and ligases, which add or remove functional groups, proteins, lipids or sugars to or from amino acid side chains and proteases, which cleave peptide bonds to remove specific sequences or regulatory subunits. Indeed, it is estimated that 5% of the proteome comprises enzymes that perform more than 200 types of post-translational modifications. PTMs occur at distinct amino acid side chains or peptide linkages, and they are most often mediated by enzymatic activity. Changes at the transcriptional and mRNA levels increase the size of the transcriptome relative to the genome, and the myriad of different post-translational modifications exponentially increases the complexity of the proteome relative to both the transcriptome and genome.Īdditionally, the human proteome is dynamic and changes in response to a legion of stimuli, and post-translational modifications are commonly employed to regulate cellular activity. While the genome comprises 20,000 to 25,000 genes, the proteome is estimated to encompass over 1 million proteins. Post-translational modifications are key mechanisms to increase proteomic diversity. PTMs are chemical modifications that play a key role in functional proteomic because they regulate activity, localization, and interaction with other cellular molecules such as proteins, nucleic acids, lipids and cofactors.
The increase in complexity from the level of the genome to the proteome is further facilitated by protein post-translational modifications (PTMs). Genomic recombination, transcription initiation at alternative promoters, differential transcription termination, and alternative splicing of the transcript are mechanisms that generate different mRNA transcripts from a single gene. These estimations demonstrate that single genes encode multiple proteins. While it is estimated that the human genome comprises between 20,000 and 25,000 genes, the total number of proteins in the human proteome is estimated at over 1 million. Within the last few decades, scientists have discovered that the human proteome is vastly more complex than the human genome.
